The Significance of the MHRA Approval and Upcoming FDA Review of the First Gene Editing Treatment
Conversations in Drug Development Trends
NOVEMBER 27, 2023
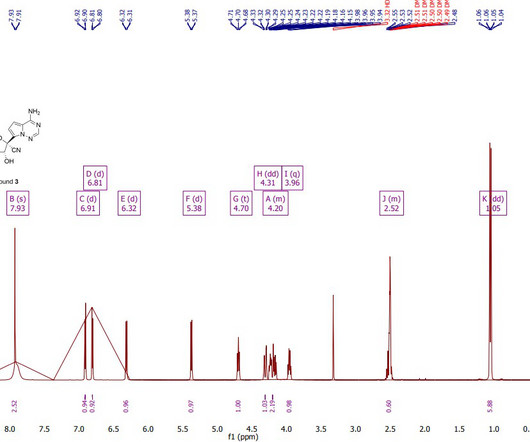
Casgevy, the commercial product formerly known as exa-cel, is administered by taking stem cells out of a patient’s bone marrow and editing a gene in the cells in a laboratory, with the modified cells then infused back into the patient after conditioning treatment to prepare the bone marrow.












































Let's personalize your content