Taking a new approach to tackle neurodegenerative diseases
Drug Discovery World
OCTOBER 4, 2022
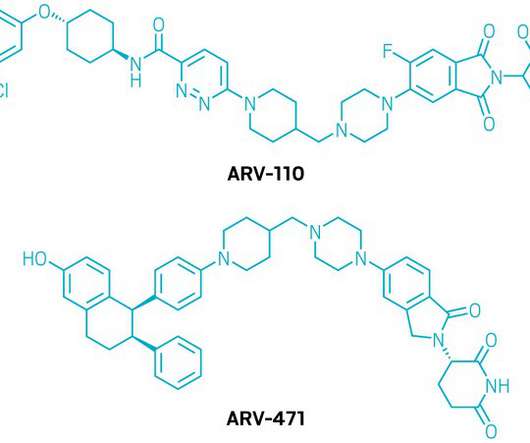
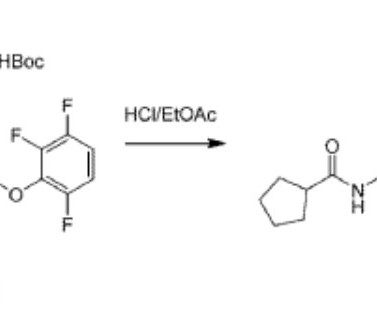
William Erhardt , President, Head of Development & Operations of biotech Oligomerix, tells Lu Rahman about the company’s differentiated approach to developing disease-modifying therapeutics for neurodegenerative diseases. . Global dementia prevalence is expected to increase to 152 million in 2050, a 204% increase compared to 2018.








































Let's personalize your content