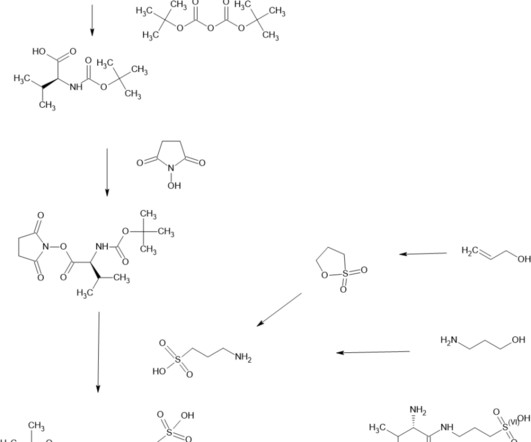
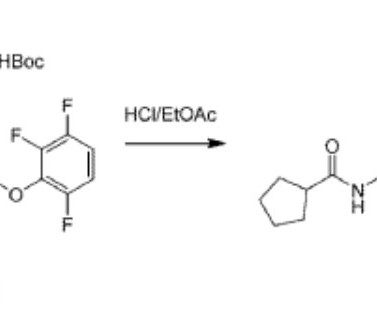
VALILTRAMIPROSATE
New Drug Approvals
OCTOBER 12, 2024
Alzheon licensed ALZ-801 from NeuroChem and is developing it for Alzheimer’s disease. 2018; 32(9): 849–861. [2]. Clinical Pharmacokinetics and Safety of ALZ-801, a Novel Prodrug of Tramiprosate in Development for the Treatment of Alzheimer’s Disease. Clin Pharmacokinet. 2018 Mar;57(3):315-333. Hey JA, et al.












Let's personalize your content