Sanofi and Translate Bio initiate Phase 1/2 clinical trial of mRNA COVID-19 vaccine candidate
The Pharma Data
MARCH 11, 2021
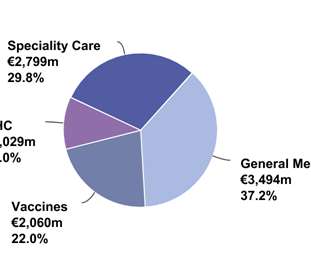
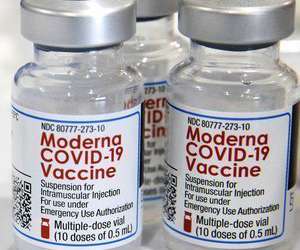
Sanofi Pasteur, the vaccines global business unit of Sanofi, and Translate Bio (NASDAQ: TBIO), a clinical-stage messenger RNA (mRNA) therapeutics company, today announced the start of the Phase 1/2 clinical trial for MRT5500, an mRNA vaccine candidate against SARS-CoV-2, the virus that causes COVID-19.




























Let's personalize your content