Researchers reprogram gene therapy viral vectors to bind specific protein targets
Broad Institute
JULY 19, 2023
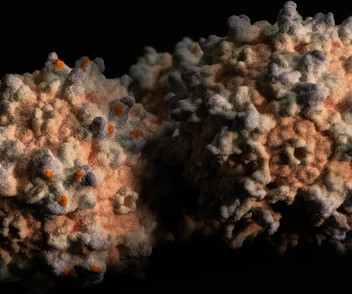
By Allessandra DiCorato July 19, 2023 Credit: Kevin Middleton, Broad Communications A three-dimensional model of adeno-associated viruses (AAVs), which scientists have engineered to package and deliver gene therapies to cells in the body. a capsid that is efficiently transported across the blood-brain barrier.


































Let's personalize your content