Roche, Adaptimmune part ways on cell therapy research
BioPharma Drive: Drug Pricing
APRIL 12, 2024
The two companies have ended a collaboration that was struck in 2021 and focused on “off-the-shelf” cell therapies for cancer.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

BioPharma Drive: Drug Pricing
APRIL 12, 2024
The two companies have ended a collaboration that was struck in 2021 and focused on “off-the-shelf” cell therapies for cancer.

BioPharma Drive: Drug Pricing
JULY 1, 2024
Like many of its cell therapy peers, Artiva, which originally sought an IPO in 2021, has shifted its strategy in hopes of riding a recent wave of investor interest in autoimmune disease research.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
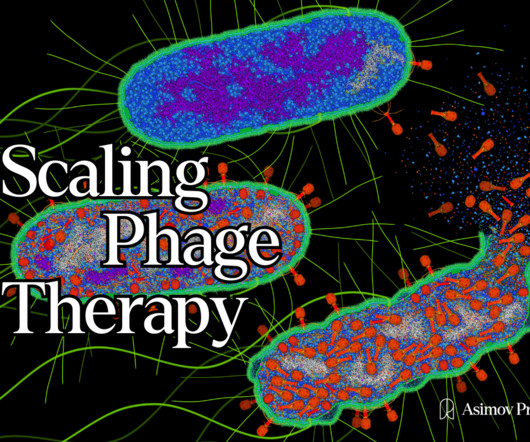
Codon
FEBRUARY 13, 2024
Tom Ireland writes about the companies and technologies that are reimagining phage therapy. Soon after its publication, scientists, journalists, and investors were revisiting ‘phage therapy’ as a promising alternative to our failing antibiotics. Read it on our website here. Illustration by David S. Fast forward to 2023.

Drug Channels
DECEMBER 19, 2021
We made it through a challenging 2021. Ring in 2022 with these noisemakers from the Drug Channels party bag: Hospitals earn incredible markups on CAR-T therapies Utilization management booms Health plans often ignore clinical guidelines Benefit designs flummox physicians Plus: an awesomely concise summary of U.S. health insurance.

FDA Law Blog: Drug Discovery
DECEMBER 7, 2022
Valentine — On November 22, 2022, FDA approved CSL Behring’s BLA for Hemgenix (etranacogene dezaparvovec), an AAV-based gene therapy for the treatment of adults with Hemophilia B who currently use Factor IX prophylaxis therapy, have current or historical life-threatening hemorrhage, or have repeated, serious spontaneous bleeding episodes.
PLOS: DNA Science
MARCH 21, 2024
The Food and Drug Administration just announced approval of Lenmeldy (atidarsagene autotemcel), a gene therapy to treat the neurological condition metachromatic leukodystrophy (MLD). The history of gene therapy for MLD is compelling – DNA Science covered it for Rare Disease Day in 2021, here. She passed away in 2013.
Drug Target Review
JUNE 19, 2024
The mission of Lineage Cell Therapeutics is to deliver on some of the early promises of cell therapy. Cell therapy as a concept is a wonderful idea, but many of the early efforts never generated the kind of clinical data that gets people excited and leads to new medicines. In some cases, cell therapy can be curative for the patient.

Drug Target Review
FEBRUARY 28, 2024
Over the past 25 years, T-cell therapies have gained significant ground in the treatment of cancer. Preclinical research on γδ T cells has made great strides since the cells were first identified in the 1980s, with γδ T-cell therapies from several companies, including IN8bio, now in or nearing clinical trials for various cancers.

Eye on FDA
JANUARY 12, 2022
During nearly all of 2021, FDA has been under the direction of an acting commissioner. COVID – 144 announcements were COVID-related in 2021, compared to 274 such announcements during 2020; Approvals – there were 77 announcements related to approvals this year, 22 of which were COVID-related announcements.

The Pharma Data
JULY 8, 2021
Nasdaq:BIIB) today announced it will report second quarter 2021 financial results Thursday, July 22, 2021, before the financial markets open. Biogen discovers, develops, and delivers worldwide innovative therapies for people living with serious neurological and neurodegenerative diseases as well as related therapeutic adjacencies.

Drug Discovery Today
JANUARY 25, 2021
OSLO/LONDON, JANUARY 20 2021: EXACT THERAPEUTICS AS (“EXACT-Tx” or the “Company”), a clinical stage precision medicine company today announced a collaboration with GE Healthcare to develop an innovative ultrasound probe to be used in forthcoming studies using Acoustic Cluster Therapy (ACT®) across multiple disease conditions.

Drug Discovery Today
MAY 11, 2021
Oslo, Norway, May 10, 2021 – Lytix Biopharma AS, a Norwegian clinical-stage immunoncology therapeutic company developing oncolytic molecule therapies to treat cancer, today announces that promising full data results from a phase I study of its lead candidate LTX-315 were published in Clinical Cancer Research, a peer-reviewed journal of the American (..)
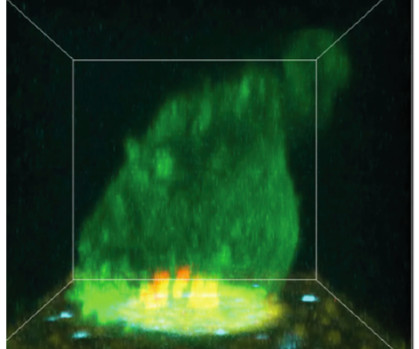
SugarCone Biotech
MARCH 24, 2025
When we look at how TCEs interact with targeted cancer cells we can quickly see how cancers respond to T Cell-based therapies. The most common response is quite expected, which is that the cancer cells escape from TCE therapy by downregulating expression of the targeted cancer protein (Ref. doi: 10.1136/jitc-2021-004348 6.
Drug Target Review
JULY 4, 2023
These therapies have broadened treatment options for patients to expand beyond the more traditional small molecule drug alternatives. Patients and caregivers also assess the benefits offered by different therapies, weighing the progression-free survival with their off-target effects.

The Pharma Data
AUGUST 3, 2021
Revenue in the second quarter of 2021 increased 23 percent, driven by volume growth of 22 percent. – Revenue from all key products grew in the quarter and 2021 year-to-date. . – Revenue from all key products grew in the quarter and 2021 year-to-date. – 2021 EPS guidance updated to be in the range of $6.73

The Pharma Data
JULY 29, 2021
econd Quarter 2021 Product Sales Increased 21% Year-Over-Year Primarily Driven by Veklury. Nasdaq: GILD) announced today its results of operations for the second quarter 2021. “We Second Quarter 2021 Financial Results. Total second quarter 2021 revenue of $6.2 As of June 30, 2021, Gilead had $7.4

PLOS: DNA Science
OCTOBER 12, 2023
In the final chapter of my 2012 book The Forever Fix: Gene Therapy and the Boy Who Saved It , I predicted that the technology would soon expand well beyond the rare disease world. Gene therapy clearly hasn’t had a major impact on health care, offering extremely expensive treatments for a few individuals with rare diseases.

Broad Institute
JUNE 10, 2024
The advance, from the lab of Broad core institute member David Liu , could one day help researchers develop a single gene therapy for diseases such as cystic fibrosis that are caused by one of hundreds or thousands of different mutations in a gene. Tags: Gene editing Gene therapy David Liu Nature Biomedical Engineering.

The Pharma Data
AUGUST 1, 2021
to $6.14; Raises 2021 Adjusted Diluted EPS Guidance Range from $12.37 Based upon the momentum of our business, we are raising our full year 2021 EPS guidance and believe AbbVie is very well positioned for the long term.” ” Second-Quarter Results. Worldwide net revenues were $13.959 billion, an increase of 33.9

KIF1A
APRIL 25, 2024
She is scheduled to be the 2nd patient in the world to receive ASO therapy for KAND. Timeline: December 2021 – We met with Dr Chung in NYC and discussed and applied to n-Lorem. March 2023 – We met with n-Lorem and found out Susannah’s ASO (1st KAND patient to receive ASO therapy for KAND) would work for Sloane.

Drug Channels
SEPTEMBER 17, 2021
Vicki discusses the advantages of integrated technology solutions in enhancing specialty patients' access to therapy, affordability, and adherence. Click here to sign up for complimentary early access to the specialty edition of the CoverMyMeds 2021 Medication Access Report. Read on for Vicki’s insights.

Advarra
NOVEMBER 29, 2022
The field of cell and gene therapies (CGT) is constantly evolving, and there has been significant progress in this area of research. However, despite the promise of these therapies, the regulations governing them lag the science, which in turn hinders the clinical translation of these novel medicines.

The Pharma Data
OCTOBER 14, 2021
The results, which demonstrate that patients treated with Biogen’s portfolio of MS therapies mount an effective antibody response to COVID-19 vaccination, are being presented at the 37th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) virtual meeting, October 13-15, 2021.

The Pharma Data
AUGUST 7, 2021
In June 2021, Wegovy , semaglutide 2.4 For the 2021 outlook, sales growth is now expected to be 10-13% at CER (previously 6-10%), and operating profit growth is now expected to be 9-12% at CER (previously 5-9%). ?? Lars Fruergaard Jørgensen, president and CEO: “We are pleased with the sales growth in the first half of 2021.

Drug Target Review
JULY 11, 2024
To date, there are less than 30 known surface proteins that form the basis of all approved cancer-targeted therapies of all modalities, including antibody-drug conjugates (ADCs), T-cell engagers, CAR T-cells and radiopharmaceuticals. These could be targets for antibody-drug conjugates (ADCs), bi-specific antibodies, or other therapies.

Advarra
JUNE 13, 2023
Research in gene therapies and genetically engineered drugs and vaccines are growing exponentially, and will only continue to become more popular. The accelerating gene therapy market is expected to grow globally by 16.6% As more therapies are moving out of the lab and into human clinical trials, this paradigm is changing.

FDA Law Blog: Drug Discovery
NOVEMBER 2, 2023
Lewis, Senior Regulatory Device & Biologics Expert — On October 20, 2023, FDA announced the availability of the final guidance authored by CBER titled “Voluntary Consensus Standards Recognition Program for Regenerative Medicine Therapies.” It finalized a draft guidance published in 2022.

Drug Channels
OCTOBER 1, 2021
David discusses how electronic prior authorization (ePA) and first-fill, buy-down programs can boost patient adherence to specialty therapies. Read more » Copyright © 2006-2021 Pembroke Consulting, Inc. On November 3rd at 1:00 pm ET, ConnectiveRx will host First Fill, No Wait: How to Make it Happen , a free webinar about copay ePA.

Drug Target Review
SEPTEMBER 25, 2024
Most targeted cancer therapies used today operate by inhibiting targets along well-known oncogenic signalling cascades. The reactivation of oncogenic signalling upstream or downstream of the driving oncogene is a well-studied source of resistance to targeted cancer therapies.

DrugBank
AUGUST 9, 2024
reported in 2021. In 2021, an estimated 537 million adults were living with diabetes, a number projected to reach 643 million by 2030. billion in 2021, is projected to reach 231.7 These new therapies have demonstrated significant LDL cholesterol reductions, but their high cost remains a barrier to widespread adoption.

Eye on FDA
AUGUST 4, 2021
Below is a chart that looks at 3 numbers – the number of AdComms; the number of new molecular entities approved each year, and the number of drugs that had breakthrough therapy designation. That said, BT would be a lagging indicator since it can be granted so early in the development process. Fewer meetings – more approvals.

The Pharma Data
AUGUST 3, 2021
In the first half of 2021, Boehringer Ingelheim has supplied more humans and animals worldwide with innovative medicines than ever before. Our strong R&D pipeline has made further, decisive progress in the first half of 2021. These medical successes have the potential to improve the lives of millions of patients worldwide.”.

Chemical Biology and Drug Design
OCTOBER 10, 2023
million people worldwide were living with HIV in 2021. This study focuses on NRTIs and NNRTIs, their binding sites, mechanisms of action, FDA-approved drugs and drugs in clinical trials, their resistance and adverse effects, their molecular docking studies, and highly active antiretroviral therapy (HAART).
The Pharma Data
SEPTEMBER 11, 2021
Food and Drug Administration (FDA) has granted Breakthrough Therapy Designation for empagliflozin as an investigational treatment for adults with heart failure with preserved ejection fraction (HFpEF), Boehringer Ingelheim and Eli Lilly and Company (NYSE: LLY) announced. o Completion: 2021. o Number of patients: 3,730.

Drug Target Review
DECEMBER 16, 2024
I envision a future where technological advances will allow us to better model clinical trials through improved patient selection, run studies in a much shorter period, and find ways to identify effective therapies sooner, so we can bring new treatments to the right patients more quickly.

The Pharma Data
JULY 6, 2021
Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new data from its haemophilia A clinical programme will be presented at the virtual International Society on Thrombosis and Haemostasis (ISTH) 2021 Congress, from 17-21 July 2021. Roche’s Chief Medical Officer and Head of Global Product Development.

Drug Target Review
OCTOBER 4, 2023
Dual payload ADCs As effective as therapies have been in treating solid and haematological cancers, tumour heterogeneity and resistance remain major clinical challenges. To overcome these obstacles, combination therapy, which delivers multiple small molecules, has emerged as a potential solution. Biomaterials 273 , 120817 (2021).

Drug Channels
AUGUST 20, 2021
Ian discusses how mobile platforms like mobileCare Manager can boost patient adherence to specialty therapies. To learn more, register for ConnectiveRx's free panel discussion on September 15, 2021, at 1:00 p.m. Read more » Copyright © 2006-2021 Pembroke Consulting, Inc. Email inquiries@ConnectiveRx.com with any questions.
The Pharma Data
SEPTEMBER 27, 2021
Food and Drug Administration (FDA) has granted Breakthrough Therapy Designation for empagliflozin as an investigational treatment for adults with coronary failure with preserved ejection fraction (HFpEF), Boehringer Ingelheim and Eli Lilly and Company (NYSE: LLY) announced. 2,3 a number one explanation for hospitalization within the U.S.

The Pharma Data
AUGUST 5, 2021
The transaction was announced on July 19, 2021. The acquisition strengthens BioNTech’s cell therapy pipeline by accelerating the individualized solid tumor Neoantigen TCR cell therapy research and development program. Kite’s singular focus is cell therapy to treat and potentially cure cancer.

Drug Target Review
OCTOBER 26, 2023
Genome engineering and gene therapies that manipulate DNA sequences in cells have driven a biotechnological revolution over the past decade. In 2021, he assumed the role of Director in Protein Therapeutics at Gilead, driving biologics to prominence. Molecular Therapy 20 , 1831-1832 (2012). Nature 578 , 229-236 (2020).
The Pharma Data
SEPTEMBER 8, 2021
today announced that data reinforcing the potential of immunotherapies to impact clinical outcomes for patients will be featured at the European Society for Medical Oncology (ESMO) 2021 Virtual Congress from September 16-21. Summary of Presentations. All times noted are Central European Summer Time (CEST). Abstract Title. Presentation.
Drug Target Review
AUGUST 24, 2023
Neutralising GDF-15 with CatalYm’s anti-GDF-15 antibody visugromab was shown to reverse its inhibitory effects and to re-sensitise tumours to anti-PD-1 treatment, achieving commensurate survival benefit of anti-GDF-15-anti-PD-1 combination therapy in vivo. 2021 Jul 6;120(13):2644-56. 2021 Dec 1;9(12):1425-38.

The Pharma Data
MAY 26, 2023
State-of-the-art cryogenic freezers alongside material transfer equipment have been installed that are designed to retain the integrity of investigational advanced therapy products by minimizing their time-out-of-environment.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content