Planning for Patient Recruitment in 2025: Strategies to Stay Ahead of the Curve
Antidote
NOVEMBER 15, 2024
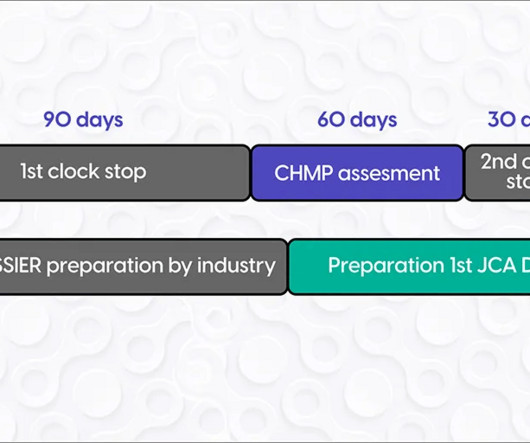
Looking toward 2025, clinical trial sponsors must plan now and rethink their patient recruitment strategies to stay ahead of evolving technologies, regulations, and shifting patient expectations.













































Let's personalize your content