Recorded Webinar: Streamlining Collaborations and Drug Development using CDD Vault
Collaborative Drug
AUGUST 1, 2023
Streamlining Collaborations and Drug Development using CDD Vault and ELN
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Collaborative Drug
AUGUST 1, 2023
Streamlining Collaborations and Drug Development using CDD Vault and ELN

Perficient: Drug Development
NOVEMBER 21, 2023
As I navigate this powerful framework, I’ve decided to document my experiences, insights, and newfound knowledge in a blog series. As I navigate through the various facets of Spring Boot, I aim to provide clear and insightful content that is accessible to beginners, seasoned developers, and anyone else eager to join me on this venture.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Collaborative Drug
JULY 22, 2024
Is Revolutionizing Drug Development. Holy Grail for Weight-Loss Drugs Is Sleep Apnea? DNA Discovery that Could Help Find Pancreatic Cancer Cure.

FDA Law Blog: Drug Discovery
NOVEMBER 10, 2024
These studies are crucial because understanding the natural course of a disease helps in designing better clinical trials and defining meaningful endpoints for future drug development. The rest of this blog will focus on the Clinical Trials Grants Program.

FDA Law Blog: Drug Discovery
MARCH 6, 2024
FDA Law Blog readers can use discount code S10-866-866L24.S Kurt provides regulatory counsel to pharmaceutical manufacturers on Hatch-Waxman patent and exclusivity, drug development, pediatric testing, and orphan drugs. More information about the conference can be found here. S for reduced registration fees.
DS in Pharmatics
SEPTEMBER 26, 2024
Discover key strategies for mastering CMC in drug development from industry leaders. Our blog covers expert insights on early-stage de-risking, CMO selection, and leveraging AI to drive success.

Plenge Gen
JANUARY 7, 2025
When I last wrote about AI on this blog three years ago, I spoke of it being a tool with the potential to transform scientific discovery, but the application I described was primarily theoretical. … The post AI Unleashed: Transforming Drug Discovery from Theory to Practice appeared first on Plenge Gen @rplenge.

FDA Law Blog: Drug Discovery
JANUARY 30, 2025
Valentine We recently blogged about a new December 2024 draft guidance about accelerated approval (the December 2024 draft guidance). This blog post focuses on interpreting these new authorities with respect to timely conduct of confirmatory trials. Tobolowsky & Charles G. Raver & James E.

addgene Blog
MARCH 21, 2024
Seeing inside these specimens has offered illumination on biochemical processes that are crucial in the world of biological research, medical therapy, and even drug development (Grimm & Lavis, 2022). If you’re struggling to select the right fluorescent tools, this blog is here to help.

The Premier Consulting Blog
JULY 31, 2024
In this blog, we explain the role of clinical pharmacology in drug development and demonstrate how the right strategy can accelerate development under the US Food and Drug Administration (FDA) 505(b)(1) and 505(b)(2) New Drug Application (NDA) pathways.

DS in Pharmatics
JULY 25, 2024
The costs of developing a new medication can amount to over $1 billion, depending on the complexity of the drug and expenses associated with unsuccessful studies. With these costs in mind, an optimized regulatory process is required to ensure that the procedures behind drug development are secure, safety-oriented, and reliable. […]

DS in Pharmatics
MARCH 22, 2024
The FDA Fast Track and Accelerated Approval pathways can expedite the review of new drugs. It is important to understand these pathways and how they impact drug development, as well as other industries, like healthcare and pharmaceutics.

Predictive Oncology
AUGUST 8, 2024
By combining these two assets, this offering represents an opportunity to de-risk drug discovery and accelerate pipeline development. The post Increasing the probability of technical success in drug development using AI and patient heterogeneity appeared first on Predictive Oncology.
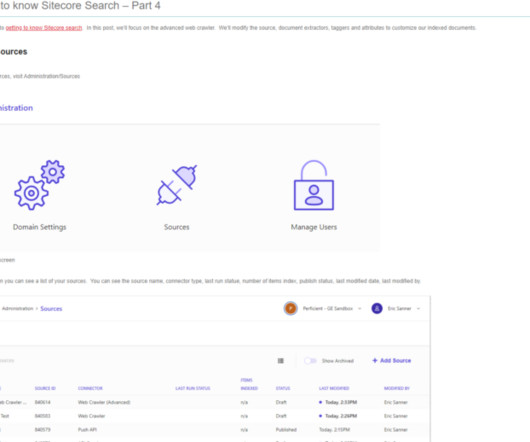
Perficient: Drug Development
JUNE 26, 2023
I did not add all the necessary fields for an actual blog article (date, author, etc). In our example, we start by importing categories then importing the blogs. When I import the blogs, I can simply join the category array from Merlin with a vertical bar ‘|’ to store in Sitecore. Merlin gave each category a GUID.

FDA Law Blog: Biosimilars
APRIL 1, 2025
While much may be in flux, the central tenet of drug development remains the same, develop drugs that are safe and effective drugs for patients, through streamlined, efficient, and cost-effective development programs. Therein lies the opportunity.

The ChEMBL-og
DECEMBER 13, 2023
A set of well-defined and structured assay descriptions would be valuable for the drug discovery community, particularly for text mining and NLP projects. This Blog post will consider the features of the 'perfect' assay description and provide a guide for depositors on the submission of high quality data. Q uestions?

PPD
JULY 29, 2024
In today’s data-driven world, AI has become valuable and indispensable, enabling organizations to extract valuable insights from vast amounts of data, make informed decisions and drive innovation across different sectors — including drug development.

FDA Law Blog: Biosimilars
JUNE 27, 2023
James is recognized as helping fundamentally shift the culture to consider the patient voice as an integral part of drug development. Mr. Karst, a co-author of the FDA Law Blog, provides regulatory counsel to pharmaceutical manufacturers on Hatch-Waxman patent and exclusivity, drug development, pediatric testing, and orphan drugs.

Reprocell
OCTOBER 3, 2024
This blog is the second in a series exploring the critical role of safety pharmacology in drug discovery. In our previous post, we discussed the importance of human tissue studies in ensuring drug safety.

Perficient: Drug Development
JULY 2, 2024
In this blog post, we will show you how to configure ServiceNow knowledge articles as a data source for Microsoft Search using the Microsoft Graph connector. We hope this blog post was helpful and you learned how to configure ServiceNow knowledge articles for Microsoft Graph Connector.

Perficient: Drug Development
FEBRUARY 27, 2025
In this blog, we’ll outline the key steps and best practices for using Groovy scripts to automate content updates. MoreAEM Insights Don’t miss out on more AEM insights and follow our Adobe blog ! However, manual updates, like rolling out a new template, can become tedious and costly when dealing with thousands of pages.

Perficient: Drug Development
MAY 30, 2024
In this blog post, I want to focus on a different quote with the same intent. When Quantity Goes Wrong I wrote another blog post on lessons learned while managing corporate innovation. Pablo Picasso said, “Inspiration exists, but it has to find you working.” Have a hundred ideas and throw away the bad ones. You’ll drown.
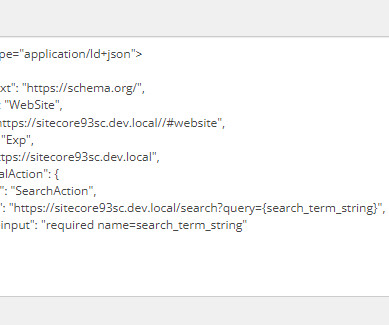
Perficient: Drug Development
OCTOBER 3, 2023
In this blog, we will be discussing Schema.org, the role of schema.org in SEO, and how to implement Schema.org in Sitecore. In the next blog, we will discuss these components in depth. What is Schema.org? Schema.org is a universal way to describe web pages with structured data. Thanks, and Stay Tuned.

Perficient: Drug Development
JUNE 13, 2023
This makes it easier to break the site into pages that have similar DOM layouts (ie: blogs, news, products, etc) for the generate command. The entity_type option sets the name of the output file “blog_site_structure” creates url list file called “crawled-urls-blogs_site_structure_default.yml”.

ACTO
DECEMBER 17, 2024
Part 2 Our first blog covered the ABCs of Artificial Intelligence (AI) and AIs evolution. Such cross-functional collaboration ensures consistent implementation from drug development through commercialization. The stakes for AI adoption in the pharmaceutical industry have never been higher.

Perficient: Drug Development
SEPTEMBER 11, 2023
I have always enjoyed writing blogs and sharing my knowledge with others. This is my 100 th blog post! Series I write a lot of blog series. Until then, I’ve shared my screen reader blog and video with many people to show them how to use a screen reader as a native user would. I can’t believe I finally made it!

FDA Law Blog: Drug Discovery
MARCH 3, 2025
FDA Law Blog is a conference media partner. Hyman, Phelps & McNamara, P.C.s As such, we can offer our readers a special 10% discount. The discount code is: D10-999-FDA25. You can access the conference brochure and sign up for the event here.
Perficient: Drug Development
OCTOBER 20, 2023
This concept is demonstrated in the Alloy Project and has been explained further in the blog post ‘ Episerver and Alternate Text for Images in the TinyMCE Rich Text Editor ‘ by Dylan McCurry. I hoped blog has helped you create a more accessible site and a better CMS experience!

FDA Law Blog: Drug Discovery
JULY 17, 2024
As originally proposed, this new Committee could be convened in conjunction with the existing medical specialty area-focused advisory committees, akin to the Drug Safety and Risk Management Advisory Committee. those reviewed by the CDER Division of Rare Diseases and Medical Genetics).

Perficient: Drug Development
AUGUST 14, 2023
It can help API developers obtain a qualitative idea of how well the API performs under real-world scenarios. In this blog, I will delve into the comprehensive process of API testing using JMeter. Conclusion: Throughout this blog, we have explored API load testing with JMeter, learning how to set up test plans and analyze test results.

Perficient: Drug Development
FEBRUARY 28, 2024
What to Expect Complimentary lunch Hear how Morgan Stanley supercharged its customer journey An opportunity to connect with peers who share your passion for digital excellence in the financial services industry before heading off for afternoon sessions at Adobe Summit Your Adobe Partner Learn more about supercharging the customer journey and delivering (..)

Perficient: Drug Development
SEPTEMBER 20, 2023
Hope that the above blog will help you to decide the lifetime of your services in Sitecore. Happy Coding

Perficient: Drug Development
JULY 6, 2023
This blog contains examples of how to obtain the AEM Component’s Style System configuration. Conclusion: In this blog, we explored the implementation of custom styles using the AEM Style System for both pages and components. Note: I utilized componentcolor.css file classes in that styles tab.

Perficient: Drug Development
MARCH 12, 2024
Looking Ahead We are excited about the new opportunities the Government Specialization brings and are committed to continuing our journey of innovation and excellence. For more information about Perficient’s Pega practice and our intelligent automation solutions, subscribe to Perficient’s blog, and follow us on Twitter and LinkedIn.

FDA Law Blog: Drug Discovery
MARCH 13, 2023
Bauer, Senior Regulatory Drug Expert & James E. Valentine — Incorporating patient and caregiver experiences into every phase of drug development has become increasingly prioritized during both development and review ( see, e.g. , previous coverage here ). By Larry J.

Perficient: Drug Development
FEBRUARY 13, 2025
Both are designed to enhance web development efficiency and performance, but they cater to different needs and use cases. In this blog, well explore the strengths, features, and considerations of each framework to help you make an informed decision. Best for: Blog posts, marketing pages, documentation. What is Remix?

Perficient: Drug Development
MAY 1, 2024
Check out our blogs for more insights from our HCL team. We are excited to see what opportunities lie ahead and look forward to supporting HCL every step of the way. Congratulations to our team on this exciting award! Need help with your HCL implementation or commerce strategy? Reach out.

Perficient: Drug Development
DECEMBER 26, 2023
Storytelling As I mentioned in my blog post about the Psychology of Innovation , we humans are wired for storytelling. In fact, in my blog post about Getting Unstuck When You’re Not the Creative Type , this concept of lateral thinking works everywhere! So why is it that many adults downplay whimsy as childish? market size (6.9%

PPD
AUGUST 28, 2024
For pharmaceutical innovators and drug developers working to bring oncology therapies to market, patients are the “why” behind it all. Ultimately, a smooth trial process paves the way for the development of promising therapies that will continue to help more patients.

Perficient: Drug Development
AUGUST 28, 2024
And stay tuned to our Salesforce blog for all our post-conference insights. Visit #PerficientDreamforce2024 to learn more. Can’t Make it to Dreamforce? Don’t worry! Schedule some time with us and let our experts fill you in.

Perficient: Drug Development
SEPTEMBER 26, 2023
These blogs delve deep into the core principles and benefits of Composable Architecture, making them a must-read for any business aiming to thrive in the digital age. Take a look below!

Alta Sciences
JULY 18, 2024
Issue 38 of The Altascientist explores the key considerations for successful formulation development and manufacture for safety assessment, alongside examining the strategies Altasciences employs to support you in planning your program. Image blog-thumbnail-issue-38-quick-hitter_0.png

BMG Labtech
MARCH 11, 2024
They are often the method of choice to detect or measure specific biological molecules (analytes) for diagnostics, drug discovery or fundamental research. Enzyme-Linked Immunosorbent Assays (ELISAs) are an essential technique in today’s laboratory with many applications in the life sciences.

Perficient: Drug Development
MARCH 4, 2025
Stay tuned for more insightful blogs! The next time you write an Apex class that interacts with sensitive fields, remember to check field level security before exposing data! Want to learn more about Salesforce security best practices?
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content