Redefining Acceleration of the Drug Development Journey
PPD
NOVEMBER 11, 2024
Where companies may otherwise face multiple hand-offs and inefficiencies, the right partner can reduce transfers, minimize risk and avoid potential errors and delays.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

PPD
NOVEMBER 11, 2024
Where companies may otherwise face multiple hand-offs and inefficiencies, the right partner can reduce transfers, minimize risk and avoid potential errors and delays.

Broad Institute
NOVEMBER 2, 2023
Bayer and Broad Institute extend cancer therapy research collaboration By Corie Lok November 2, 2023 Breadcrumb Home Bayer and Broad Institute extend cancer therapy research collaboration Long-standing industry-academia collaboration has already resulted in three clinical oncology candidates.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
The Pharma Data
JULY 29, 2021
Loxo Oncology at Lilly, a research and development group of Eli Lilly and Company (NYSE: LLY), and Kumquat Biosciences today announced an exclusive collaboration focused on the discovery, development and commercialization of potential novel small molecules that stimulate tumor-specific immune responses.

Drug Target Review
OCTOBER 11, 2023
Autophagy in a hot topic in scientific circles; the number of papers that mention the process has risen almost exponentially since Yoshinori Ohsumi was awarded the Nobel Prize in 2016 for his research into its underlying mechanisms. Autophagy boosters, in contrast, are conventional small molecule drugs. cited 2023 Sep 5].

The Pharma Data
NOVEMBER 19, 2020
The Company is also aiming to leverage the highly conserved structure of the SARS-CoV-2 M pro protease as a basis for the design of novel oral small molecules against predicted future variants of SARS-CoV-2 and other related human viruses. References. Hilgenfeld, Febs J. 2014), 281, 4085-4096. Pillaiyar, et al, J.

LifeSciVC
JANUARY 16, 2024
The market for each target class likely asymptotes with the number of Pharma or large biotech who can clinically develop and commercialize such assets; thus, there is intense focus on the first handful of assets to market. Small molecule GLP1s?

The Pharma Data
NOVEMBER 30, 2020
Biohaven acquires exclusive global rights to a portfolio of novel, small-molecule CGRP antagonists. The lead candidate, HTL0022562, has advanced through preclinical development demonstrating promising and differentiated properties for further investigation in human trials. Vlad Coric , M.D.,

The Pharma Data
APRIL 6, 2021
Bayer will present new research across its oncology portfolio at the virtual American Association for Cancer Research (AACR) Annual Meeting 2021 , taking place over two weeks on April 10-15 and May 17-21, 2021. Overall, these data stress Bayer’s commitment to continued research in some of the company’s key areas of focus in oncology.

Conversations in Drug Development Trends
MAY 30, 2024
These advancements have fostered increased focus from politicians, policymakers, and the investment communities backing academic and industry-sponsored research projects. Differences in regulatory sentiments and industry sponsors’ subsequent clinical development strategies historically restricted access based on geography.

Drug Target Review
JULY 18, 2023
AI is being used to find the targets themselves, design the drugs to manipulate that biology, and thirdly, AI is being used to support validation of those targets and drugs as part of pre-clinical development. There is a plethora of data available to researchers, but it is not all standardised and lacks the same metadata.

The Pharma Data
NOVEMBER 18, 2021
— Gilead Exercises Options to Arcus’s Anti-TIGIT Program (Domvanalimab and AB308), Etrumadenant (A2a/A2b Adenosine Receptor Antagonist) and Quemliclustat (Small Molecule CD73 Inhibitor) — — Arcus to Receive Option Payments Totaling $725 million — Gilead Lores,Inc. Nasdaq GILD) and Arcus Biosciences,Inc.
Nvidia Developer: Drug Discovery
MARCH 21, 2023
New supercomputing-scale large language models (LLMs) that understand biology and chemistry text are helping scientists understand proteins, small molecules, DNA, and biomedical text. These state-of-the-art AI models help generate de novo proteins and molecules and predict the 3D structures of proteins.
Drug Target Review
MAY 1, 2024
The advent of monoclonal antibodies paired with improvements in each ADC design component has led to the approval of 11 ADCs with more than 180 ADCs currently in clinical development. He has more than 25 years of US-based experience in biologics drug discovery and development in biotechnology research.

LifeSciVC
APRIL 2, 2024
We are turning a corner, and optimistically that means more private companies will be able to secure the resources they need to advance groundbreaking research through critical value inflection points. We experienced strong enthusiasm for small molecule research in the investor community. What resonated?

The Pharma Data
NOVEMBER 11, 2020
“These data provide further evidence of efficacy and high potency of SLV213 against SARS-CoV-2 and support the clinical development of SLV213 as a potential oral treatment for COVID-19,” said Ted Daley, President and CEO, Selva Therapeutics. “As About SLV213. About Selva Therapeutics.
Drug Target Review
DECEMBER 13, 2024
7,8 The discovery of these biomarkers provides valuable insights into prognosis and disease progression, while also guiding the clinical development of new targeted immunotherapies. However, research and clinical trials of BiTEs in solid tumours, including NSCLC, are still in the early phases. Biomarker Research.

The Pharma Data
MARCH 28, 2022
Our Research & Development (R&D) teams are following the science to control chronic inflammation and collaborating with leading experts across all sectors to address both urgent and growing patient needs. Two complementary candidates for COPD , developed in collaboration with Regeneron, targeting distinct subpopulations.

The Pharma Data
JANUARY 13, 2021
Knopp Biosciences is a privately held drug discovery and development company focused on delivering breakthrough treatments for immunological and neurological diseases of high unmet need. Knopp’s clinical-stage oral small molecule, dexpramipexole, is in Phase 2 clinical trials in moderate-to-severe eosinophilic asthma.

The Pharma Data
SEPTEMBER 25, 2020
ICR and Cancer Research UK spinout closes Series B financing. Monte Rosa Therapeutics has raised $96m in Series B financing to support further develop of its pipeline of small-molecule protein degraders. Source link.

The Pharma Data
JUNE 28, 2021
Broadens company’s oncology platform of Targeted Alpha Therapies / Acquisition includes actinium-225 labeled differentiated PSMA small molecule for the treatment of prostate cancer. Xofigo is currently under further evaluation in a broad clinical development program in prostate cancer and beyond.

Drug Target Review
JULY 18, 2024
Beyond the standard tumour-informed assay, we collaborated with Natera to create a more sensitive tumour-informed test using its Signatera molecular residual disease testing technology for research use in our AMPLIFY-201 clinical trial. This helped us to identify as many patients as possible who already had microscopic tumour spread.

The Pharma Data
DECEMBER 10, 2020
executive vice president of research and development. “We We look forward to working closely with the FDA throughout the clinical development process to bring this potential new innovative treatment to patients as quickly as possible.”. About LX9211. Safe Harbor Statement.

Drug Target Review
JULY 11, 2023
In the quest to combat and eradicate diseases, scientists and researchers are increasingly turning to the immense potential of artificial intelligence (AI) as a tool. Her focus initially consisted of preclinical development strategies , clinical development optimisation, target discovery and validation.

The Pharma Data
MAY 31, 2022
We look forward to continuing our collaboration with Biogen and the Parkinson’s community in our unified goal to develop BIIB122 as a potential treatment option for people and families living with Parkinson’s disease.”. BIIB122 is an investigational small molecule inhibitor of LRRK2 that was discovered and developed by Denali.

The Pharma Data
OCTOBER 27, 2020
Mirati Therapeutics is a San Diego -based late-stage biotechnology company relentlessly focused on translating drug discovery and research into new treatments for patients by advancing and delivering novel therapeutics that target the genetic and immunologic drivers of cancer. About Mirati Therapeutics.

The Pharma Data
APRIL 11, 2023
Bayer is progressing novel research around its prostate cancer treatment darolutamide. Both small molecule inhibitors in Immuno-Oncology are being jointly developed in a strategic research alliance with the German Cancer Research Center (DKFZ) in Heidelberg, Germany.

The Pharma Data
DECEMBER 17, 2020
Cadavid will be responsible for the strategy, direction and execution of the company’s clinical development programs. Prior to Fulcrum, Cadavid held several leadership positions at Biogen, including Senior Medical Director of the multiple sclerosis clinical development group. He spent 27 years at Merck & Co.

The Pharma Data
OCTOBER 18, 2020
Dr. Berk, commented “I am excited to join the Board of Inflection Biosciences as it advances its novel PIM/PI3K inhibitor, IBL-202, towards clinical development for unmet needs in B-cell malignancies and other cancers.”. Dr. Berk most recently has served as a consultant to several companies developing oncology therapies.

The Pharma Data
MAY 15, 2023
The acquisition complements Gilead’s existing clinical development priorities by adding additional pipeline assets for well-validated targets in oncology and inflammation. Executive Vice President, Research, Gilead Sciences. The company was founded in 2021 by Stephen Kaldor, Ph.D., Qing Dong, Ph.D., and Gene Hung, M.D.,

The Pharma Data
FEBRUARY 22, 2021
(Nasdaq: RIGL) today announced a global exclusive license agreement and strategic collaboration to co-develop and commercialize Rigel’s R552, a receptor-interacting serine/threonine-protein kinase 1 (RIPK1) inhibitor, for all indications including autoimmune and inflammatory diseases. vice president of immunology at Lilly.

Sygnature Discovery
NOVEMBER 29, 2023
QN-302 has been conceived to work by selectively stabilizing G-quadruplex complexes prevalent in the promoter region of cancer-related genes in many tumor types, impeding the function of these cancer genes, and may therefore potentially offer a tumor-selective clinical approach to treatment. Qualigen Therapeutics’ Chief Medical Officer.

The Pharma Data
OCTOBER 27, 2020
AavantiBio’s strategic partnership with University of Florida’s Powell Gene Therapy Center provide their foundational research in rare genetic disorders. Sirnaomics is the only biopharma conducting R&D and clinical development in the field of RNAi therapeutics in both the U.S. Primmune Therapeutics . The company’s $27.4
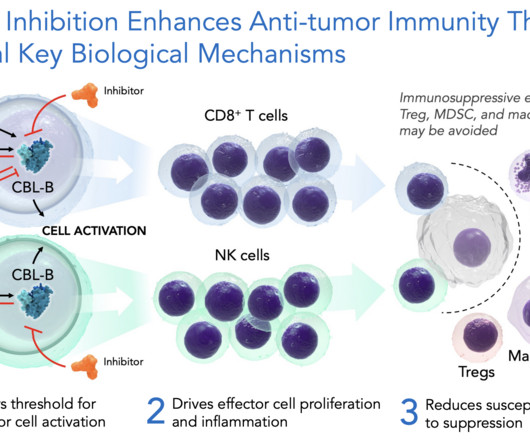
LifeSciVC
MARCH 1, 2023
Importantly, they also pointed to something that we at HotSpot Therapeutics and a small group of other researchers and companies recognized several years ago – that scientific advances have made this attractive, but once considered ‘undruggable,’ target druggable.

The Pharma Data
OCTOBER 14, 2020
Read-through therapeutic development is focused on extending mRNA half-life and increasing protein synthesis by enabling the cytoplasmic ribosome to read through premature stop codons to produce full-length proteins. ELX-02 is in the early stages of clinical development focusing on cystic fibrosis.

The Pharma Data
FEBRUARY 21, 2022
Bayer’s research and development pipeline continues to grow as the company is building on its existing competencies, such as the expertise around small molecules, while expanding into new modalities, including cell and gene therapies.

The Pharma Data
DECEMBER 21, 2020
Program focused on identifying small molecules that target a GPCR for degradation as potential therapeutic agents for gastrointestinal disorders. The principle of TPD is to use small molecules to commit the target protein into the E3 ligase-mediated degradation pathway thereby eliminating or reducing its activity.

The Pharma Data
MARCH 1, 2022
. “With AbbVie’s acquisition of Syndesi, we aim to advance the research of a novel, first-in-class asset for the potential treatment of cognitive impairment associated with neuropsychiatric and neurodegenerative disorders.”

The Pharma Data
JANUARY 10, 2021
(Nasdaq:IDYA), an oncology-focused precision medicine company committed to the discovery and development of targeted therapeutics, today announced it has submitted an Investigational New Drug (IND) application with the U.S.

The Pharma Data
DECEMBER 15, 2020
Researchers are aware of more than 80 diseases that occur when the immune system attacks the body’s own organs, tissues and cells. Many drug makers are investing in new science and hope to develop new therapeutics that address autoimmune disease. Treatment induced sustainable clinical responses and reduced systemic inflammation.

The Pharma Data
JANUARY 7, 2021
As CEO of Freeline, a liver-directed gene therapy company, she scaled the company from preclinical stage to a fully integrated biotechnology organization, which included a broad, internally developed pipeline, two programs in clinical development and a commercial-scale, high-quality CMC and manufacturing platform.

The Pharma Data
JANUARY 13, 2021
Tonix is a clinical-stage biopharmaceutical company focused on discovering, licensing, acquiring and developing small molecules and biologics to treat and prevent human disease and alleviate suffering. Tonix’s portfolio is primarily composed of central nervous system (CNS) and immunology product candidates.
Codon
MARCH 20, 2023
Nucleic Acids Research. Marine biofilm engineered to produce current in response to small molecules. Nucleic Acids Research. Nucleic Acids Research. Nucleic Acids Research. Health Single-cell transcriptomic atlas-guided development of CAR-T cells for the treatment of acute myeloid leukemia.

The Pharma Data
OCTOBER 19, 2020
The appointments of Maarten and Teun come at an essential time for AM-Pharma as we finalize all preparations to move into the last stage of clinical development for our lead program,” said Erik van den Berg , Chief Executive Officer at AM-Pharma. Dr. Kraan joins AM-Pharma from Pierre Fabre S.A., Kenilworth, N.J.,

The Pharma Data
OCTOBER 21, 2020
Research s howed KB-0742 i nhibited a ndrogen r eceptor -d ependent a ctivity and r educed t umor g rowth in c astration -r esistant p rostate c ancer. Company is on t rack to s ubmit an IND in the fourth quarter of 2020 and i nitiate a Phase 1 /2 clinical trial for advanced solid tumors in 2021. SAN MATEO, Calif.,
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content