FDA Approves First COVID-19 Treatment for Young Children
The Pharma Data
APRIL 26, 2022
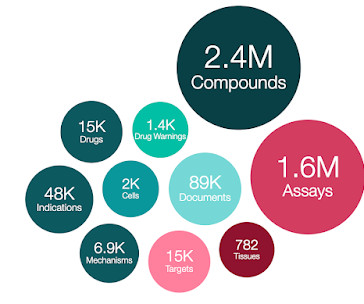
Learn more about FDA-approved and authorized COVID-19 vaccines. Given the similar course of COVID-19 disease in adults and pediatric patients, today’s approval of Veklury in certain pediatric patients is supported by efficacy results from phase 3 clinical trials in adults.
































Let's personalize your content