Shifting Paradigms in PAH Clinical Trials: 7 Key Takeaways for Success
PPD
AUGUST 5, 2024
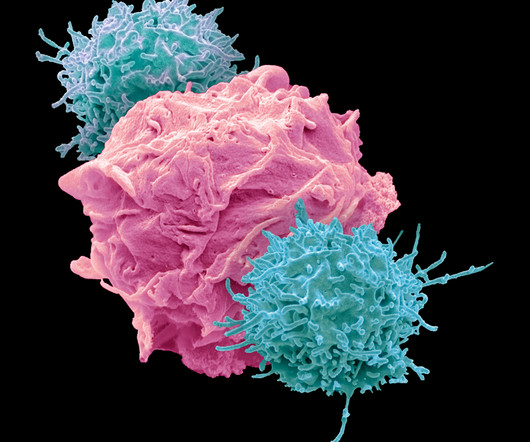
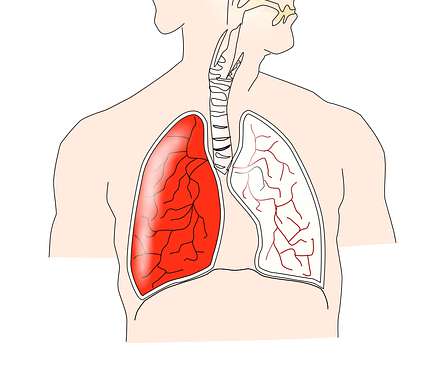
Food and Drug Administration (FDA)-approved therapies for treating PAH were primarily vasodilators, designed to overcome the imbalance between vasoactive and vasodilator mediators and to restore endothelial cell function. Their early definition — as well as plans for recording and tracking — is a major factor in a trial’s success.









































Let's personalize your content