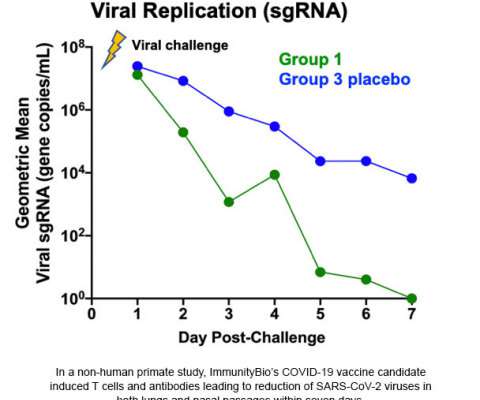
ImmunityBio’s hAd5 T-Cell COVID-19 Vaccine Candidate Shows Complete Protection of Airways in Non-Human Primates
The Pharma Data
DECEMBER 10, 2020
–( BUSINESS WIRE )– ImmunityBio , a privately-held immunotherapy company, today announced its COVID-19 vaccine candidate protected nasal and lung airways of non-human primates against coronavirus (SARS-CoV-2) in a challenge study. 11, 2020 02:48 UTC. CULVER CITY, Calif.–(























Let's personalize your content