FDA approval represents ‘transformational progress’ in hypertension
Drug Discovery World
MARCH 22, 2024
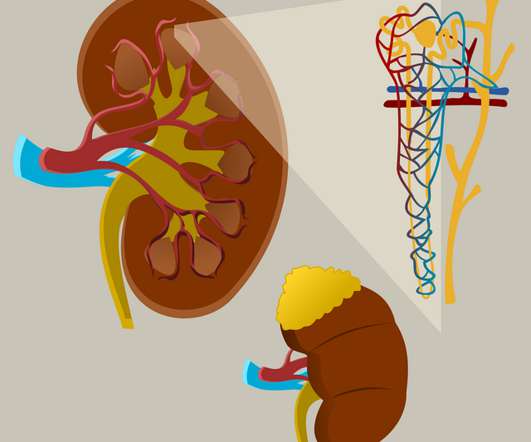
This is a major public health issue leading to a high incidence of cardio- and cerebrovascular events. ” The post FDA approval represents ‘transformational progress’ in hypertension appeared first on Drug Discovery World (DDW).











































Let's personalize your content