FDA approves Covid-19 mAb for emergency use in immunocompromised
Drug Discovery World
MARCH 26, 2024
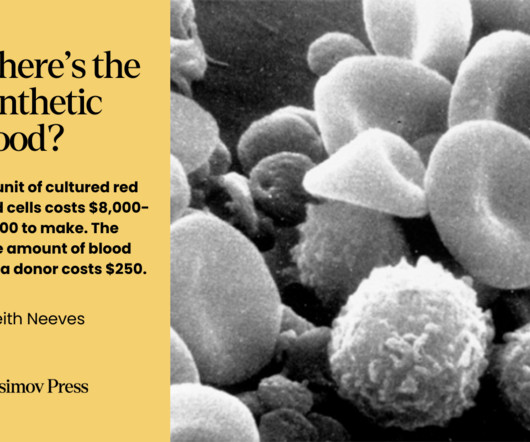
It is approved in adults and adolescents (12 years of age and older weighing at least 40kg) who are moderate-to-severely immune compromised due to certain medical conditions or receipt of certain immunosuppressive medications or treatments and are unlikely to mount an adequate immune response to Covid-19 vaccination.





































Let's personalize your content