Deliberate Dysentery
Codon
JANUARY 21, 2024
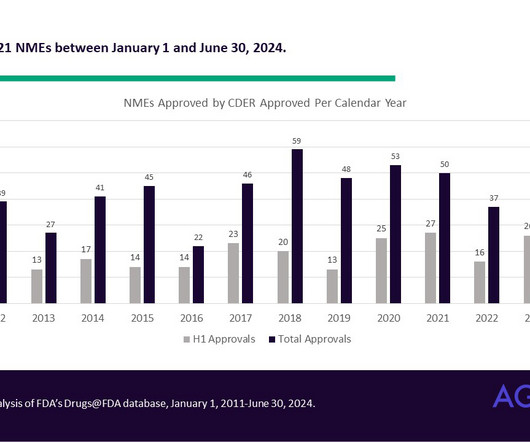
Today, refined versions of these human challenge studies have become standard practice in testing vaccines for vector-borne diseases (e.g., Human challenge trials were an indispensable part of the development of the malaria vaccine, R21/Matrix-M, endorsed by the World Health Organization last October.




































Let's personalize your content