bamlanivimab and etesevimab together for treatment of COVID-19 in the U.S.
The Pharma Data
APRIL 16, 2021
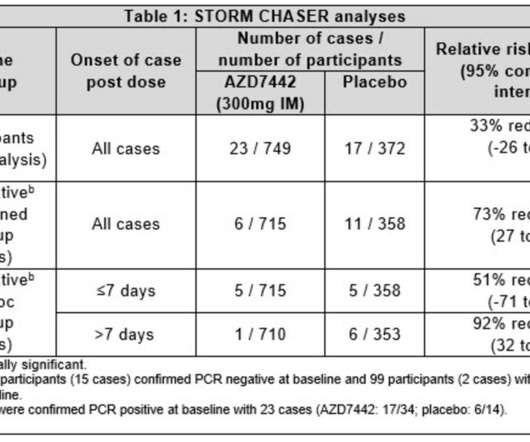
for the treatment of COVID-19 – as planned with the FDA – follows the modification of contracts with the U.S. 1.429 California strain that currently accounts for 50 percent of the virus in California and over 10 percent across a number of additional states. This request is not due to any new safety concern. All sites in the U.S.















Let's personalize your content