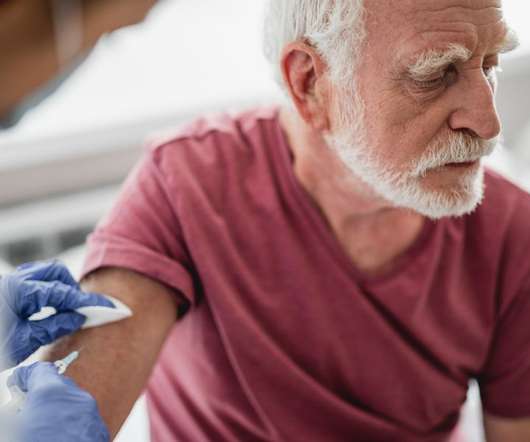
U.S. CDC Advisory Committee on Immunization Practices Recommends Vaccination with Moderna’s COVID-19 Vaccine for Persons 18 Years and Older
The Pharma Data
DECEMBER 19, 2020
Nasdaq: MRNA) a biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines today announced that the U.S. 11 ACIP members voted in favor of the vaccine and 0 members voted against. Healthcare workers have been on the front lines of the fight against the virus and are an inspiration to us all.



















Let's personalize your content