GSK antibody drug reduces COPD attacks in trial
BioPharma Drive: Drug Pricing
SEPTEMBER 6, 2024
New trial results could offer support for an expansion of Nucala’s label after U.S. regulators rejected GSK’s submission in 2018.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

BioPharma Drive: Drug Pricing
SEPTEMBER 6, 2024
New trial results could offer support for an expansion of Nucala’s label after U.S. regulators rejected GSK’s submission in 2018.

FDA Law Blog: Drug Discovery
JANUARY 30, 2025
However, as we note in that post, the design, timing of initiation, and timely conduct of confirmatory trials are also important considerations in FDAs determination of whether accelerated approval is appropriate. This blog post focuses on interpreting these new authorities with respect to timely conduct of confirmatory trials.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Target Review
DECEMBER 20, 2024
However, recent breakthroughs in AI, such as predictive modelling, clinical trial optimisation, and personalised medicine, have demonstrated its potential. We spoke with Aaron Smith, a mathematician-turned-machine learning scientist and the founder of Unlearn , a company leading the charge in applying AI to optimise clinical trial efficiency.

BioPharma Drive: Drug Pricing
MAY 10, 2024
The regulator cited “administrative constraints,” rather than any issue with Moderna’s trial data, for missing a May 12 deadline, the company said.

FDA Law Blog: Drug Discovery
NOVEMBER 10, 2024
Each year, FDA selects a limited number of clinical trials to fund to help sponsors pursue development of medical products for rare diseases and advance their field. In October, FDA announced seven new clinical trial grants awarded in fiscal year (FY) 2024 – including one for a Phase 3 trial – totaling $17.2

PPD
FEBRUARY 19, 2024
The European Union Clinical Trial Regulation (EU CTR) brings the biggest change in the regulatory landscape since the implementation of the EU Clinical Trials Directive in 2004, requiring vast changes in the way organizations are structured and conduct their day-to-day activities.

Drug Target Review
APRIL 7, 2025
A surrogate endpoint is a marker used in clinical trials as a substitute for a direct clinical outcome. For example, transcriptomic processes are showing the potential to identify and track failures in gene expression and gene regulation of amyloid and tau-related biomarkers, understood as precursors to the onset of Alzheimers disease (AD).

Drug Patent Watch
DECEMBER 9, 2024
Robust Clinical Trial Design : Clinical trials for biosimilars should be designed to demonstrate equivalence or non-inferiority to the reference product. This involves the use of statistical techniques such as equivalence and non-inferiority testing, as well as adaptive design approaches to reduce sample size and trial duration.

BioPharma Drive: Drug Pricing
AUGUST 26, 2024
Regulators in Europe granted approval based on remission rates, while the FDA sought more progress in confirmatory trials when it rejected Ordspono.

ProRelix Research
APRIL 17, 2025
andIndianregulations,ethicalconcerns,andinternationalstandardsincross-borderclinical trials. How clinicaltrialoutsourcingtoIndiaimpactsglobalpharmaresearch.ExploreU.S. Indian,andinternationalregulatorychallengesandethicalimplicationsof conductingclinicalresearchacrossborders.

ProRelix Research
JUNE 27, 2024
In the United States, The Food and Drug Administration (FDA) regulates and oversees clinical trials of food and dietary supplements and develops various regulations and guidelines for their use. The […] The post Food and Dietary Supplements Clinical Trials Regulations in USA appeared first on ProRelix Research.

Codon
APRIL 24, 2025
About one-fourth of all clinical trials and early drug development now happens in China. Chinese companies will often run the phase I trial in China for cheap, then flip it to a Western [pharmaceutical company] to run the expensive US trials and bring the drug to market." regulators hoping to speed up drug development, too.

PPD
AUGUST 16, 2023
The COVID-19 pandemic rapidly accelerated the adoption of hybrid and decentralized clinical trial (DCT) models. However, as the world settles into its post-pandemic state and returns to pre-pandemic paradigms in many areas, the pharmaceutical industry remains dedicated to moving beyond traditional, centralized clinical trial constructs.

Advarra
NOVEMBER 14, 2024
In an era where clinical trials are increasingly global, it’s more imperative than ever to leverage international expertise. Data and safety monitoring boards (DSMBs), also known as data monitoring committees (DMCs), play a critical role in overseeing a clinical trial’s safety and efficacy.

DrugBank
OCTOBER 30, 2024
From advancing drug discovery, managing clinical trials, or developing new healthcare solutions, reliable and flexible access to quality data is essential for success. From early-stage drug discovery to complex clinical trials, Snowflake scales alongside your work to meet your growing demands.

Conversations in Drug Development Trends
MAY 10, 2024
Over the past two decades, industry-sponsored clinical trials have targeted treatments for BPD, yielding some promising outcomes; however, broader psychiatric research often excludes BPD patients, a trend that extends to the emerging field of psychedelic studies.

PPD
AUGUST 5, 2024
Their early definition — as well as plans for recording and tracking — is a major factor in a trial’s success. Their early definition — as well as plans for recording and tracking — is a major factor in a trial’s success.
Advarra
JANUARY 13, 2023
New privacy regulations seem to form every few months, especially with individual U.S. states adopting their own privacy regulations (e.g., This blog explores what it takes for an EAC/CEC to adequately support worldwide clinical trials. The Nigerian Data Protection Regulation (NDPR). The Global Reach of EACs and CECs.

Agency IQ
APRIL 12, 2024
BY LAURA DIANGELO, MPH In the past few years, regulators and researchers have shown increased interest in leveraging decentralized trial methods – especially following the pandemic. In this article, AgencyIQ explores the current regulatory state of decentralized trials and what’s likely coming next.

ProRelix Research
AUGUST 3, 2023
Although the United States Food and Drug Administration (FDA) has always supported and advocated the idea of decentralized trials, the real adoption and application of decentralized aspects in trials has […] The post FDA Guidance on Advancement of Decentralized Clinical Trials appeared first on ProRelix Research.
Conversations in Drug Development Trends
AUGUST 9, 2024
Authors: Matt Cooper, PhD, Executive Director, Therapeutic Strategy Lead, Oncology; Megan Morrison, Vice President, Asia Pacific Strategy Lead Adaptive trial designs have become essential in oncology, offering a flexible and efficient approach for conducting clinical trials.

PPD
APRIL 30, 2024
31, 2022, the EU Clinical Trial Regulation (EU CTR) 536/2014 became applicable, heralding a new era for clinical trials in the European Union (EU). In addition, the constant evolution of EMA and Clinical Trials Coordination Group (CTCG) guidelines requires constant attention.

Fierce BioTech
MARCH 25, 2025
How Japan and South Korea are Shaping the Next Era of Clinical Development Discover why Japan and South Korea are emerging as clinical trial powerhouses, offering cutting-edge infrastructure, streamlined regulations, and unique commercial advantages. CMIC Group Resource Type Whitepaper CMIC_ListingLogo_250x190.png

FDA Law Blog: Drug Discovery
MARCH 7, 2024
Tobolowsky & Véronique Li, Senior Medical Device Regulation Expert & David B. Moreover, DMCs are being used in trials of modest size and in the context of increased globalization of medical product development. In another update, the recent draft guidance added “entities reviewing safety data” and adaptation committees.

Conversations in Drug Development Trends
AUGUST 23, 2023
Patients are the backbone of clinical trials, playing an essential role in the drug development process. This engagement is often less understood and is underutilized by sponsors, meaning a significant element of the trial and drug experience is missed during sponsor engagement with the FDA.
Conversations in Drug Development Trends
NOVEMBER 12, 2024
Despite their exciting potential, the smooth operation of cell therapy development trials requires extraordinary orchestration, perfectly aligning the product and patient journeys. While clinical supply is essential to any successful trial, autologous cell therapy trials occupy the far end of the spectrum regarding risk tolerance.
Conversations in Drug Development Trends
APRIL 3, 2024
Are you aware of the challenges you must address for a successful radiopharmaceutical trial? Enhancing Patient Participation in Radiopharmaceutical Trials Patient recruitment is a critical yet challenging part of radiopharmaceutical trials.

Drug Patent Watch
DECEMBER 30, 2024
Types of Drug Applications The PMDA accepts three main types of drug applications: Investigational New Drug (IND) : Required for conducting clinical trials in Japan. Challenges and Opportunities Ethnic Bridging : Japan has historically required clinical trials to be conducted within the country to account for ethnic differences.

ProRelix Research
FEBRUARY 8, 2022
A lot has changed over the past few decades in the way clinical trials are conducted. The post Clinical Trial Outsourcing and Management in USA appeared first on ProRelix Research. Advances in data management systems, risk-based monitoring, pharmacovigilance, electronic databases, and application of complex […].

PPD
MARCH 11, 2025
Breaking through research barriers Challenge #1: Small groups of patients Rare diseases impact a small number of individuals, making it difficult to recruit enough participants for clinical trials. Moreover, in some rare disease trials, it is unethical to design a control group of patients with a placebo.

Quanticate
AUGUST 19, 2024
An audit trail is a comprehensive log that records all activities, including modifications, additions, and deletions of data, within a clinical trial. It acts as a chronological record that helps trace the path of data, providing evidence that the data collected was performed in accordance with the protocol and regulatory standards.

PPD
AUGUST 28, 2024
For many patients, involvement in oncology clinical trials represents a last hope for an effective therapy. This is why oncology trials must be built around patient needs, and sponsors need to balance the complexity of oncology trials with a patient-centric mindset. Many of these patients’ conditions are disabling.

PPD
DECEMBER 16, 2024
Cost and complexity go hand-in-hand The rising costs and growing complexity in clinical trials are deeply linked, with patient recruitment, extended timelines and meeting regulatory demands emerging as some of the key drivers. Nearly half (49%) of clinical trial sponsors surveyed identified rising costs as their foremost concern in 2024.

Conversations in Drug Development Trends
DECEMBER 19, 2024
At the recent World Orphan Drug Congresses (WODC) in Europe and the United States, Worldwide Clinical Trials Derek Ansel , Vice President, Therapeutic Strategy Lead, Rare Disease, facilitated roundtable discussions to explore the operational challenges and ethical barriers surrounding genetic testing.

PPD
FEBRUARY 10, 2025
By harnessing the full range of innovative technologies and taking advantage of an FSP partners extensive skills and experience, sponsors are able to bring their therapies to market more quickly and within budget even in the face of complicated global regulations and widely fluctuating workloads.

Conversations in Drug Development Trends
MAY 1, 2024
This process can be daunting, but understanding how to manage feedback effectively is crucial for developing and ultimately gaining approval for new therapies, especially in oncology clinical trials. Additionally, consider the locations of your trial; if it is multiregional, you can start planning for your overall product approval strategy.
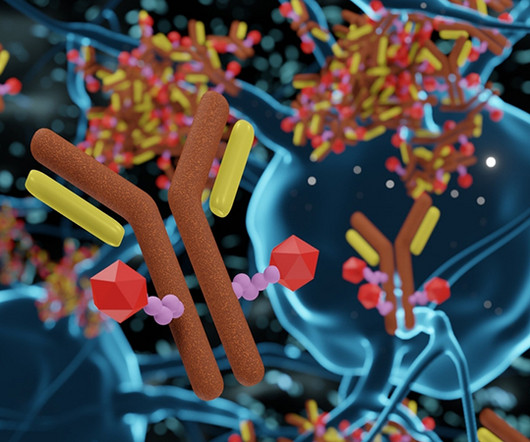
Drug Target Review
JULY 4, 2023
1 Regulators invest significant consideration balancing quality-of-life measures with overall survival when assessing novel oncology treatments. 2 However, when dosed at the MTD, ADCs display improved efficacy over small molecules in oncology trials. 3D rendering of Antibody Drug Conjugate Molecules.

thought leadership
JULY 1, 2024
Although this regulation applies to CTAs for gene therapy studies, it does not cover the additional requirement for a Genetically Modified Organism (GMO) application for such studies.

Advarra
MARCH 6, 2025
For more than 35 years, Advarra has been committed to protecting the rights and welfare of clinical trial participants while helping to improve healthcare outcomes, advancing medical knowledge, and bringing innovative, life-extending treatments to market that benefit millions of patients worldwide.

Vial
JUNE 24, 2024
Contract research organizations (CROs) and pharmaceutical companies can leverage these cutting-edge technologies to streamline clinical trials and introduce automation in drug discovery. As clinical trials grow in complexity, the volume of data being gathered and utilized for these studies is expanding.

Advarra
AUGUST 8, 2024
Artificial intelligence (AI) has taken the world by storm – and regulators are paying attention. These risks face fewer regulations but must meet certain standards to ensure ethical use. High Risk AI-powered Systems: Key Requirements The AI Act will likely consider many AI-based systems used in clinical trials today as “high risk.”
SCIENMAG: Medicine & Health
NOVEMBER 27, 2023
An innovative stem cell-based treatment for Type 1 diabetes can meaningfully regulate blood glucose levels and reduce dependence on daily insulin injections, according to new clinical trial results from the University of British Columbia (UBC) and Vancouver Coastal Health (VCH).

thought leadership
JULY 2, 2024
Change in the Submission of the Summary Notification Information Format The Summary Notification Information Format Form As of January 31, 2023, Sponsors are required to submit a Clinical Trial Application (CTA) in the EU through the Clinical Trial Information System (CTIS).

PPD
OCTOBER 17, 2024
There has also been an increase in government and regulatory support for CGT trials, in conjunction with an increase in investments for these products to get to market. Common challenges for certain patient groups include long-distance travel to clinical sites, time away from work and limited access to knowledge about CGT trials.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content